-
Entrada Therapeutics Announces Clinical Candidate, ENTR-601-45, for the Potential Treatment of People Living with Duchenne Muscular Dystrophy who are Exon 45 Skipping Amenable
来源: Nasdaq GlobeNewswire / 09 1月 2023 07:00:01 America/New_York
BOSTON, Jan. 09, 2023 (GLOBE NEWSWIRE) -- Entrada Therapeutics, Inc. (Nasdaq: TRDA), a biopharmaceutical company aiming to transform the lives of patients by establishing intracellular Endosomal Escape Vehicle (EEV™)-therapeutics as a new class of medicines, today announced the selection of a second clinical candidate within its Duchenne franchise, ENTR-601-45, for the potential treatment of people living with Duchenne who are exon 45 skipping amenable. The Company plans to submit an Investigational New Drug (IND) application in the second half of 2024.
“There is a profound unmet need for people living with Duchenne and the more than 1,300 people in the US and Europe who are exon 45 skipping amenable,” said Dipal Doshi, President and Chief Executive Officer of Entrada Therapeutics. “We believe that our portfolio of EEV-therapeutic candidates has the potential to provide a transformational impact for these individuals and their families and look forward to continuing to expand our pipeline of therapeutics for those in the Duchenne community.”
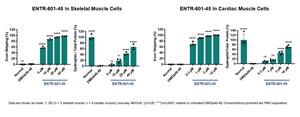
The selection of ENTR-601-45 is based on robust in vitro exon skipping and dystrophin restoration observed in patient derived skeletal and cardiac muscle cells (Figure 1) as well as in vivo preclinical data demonstrating exon skipping levels of over 90% in skeletal muscle in a hDMD mouse model (Figure 2). Entrada plans to present additional data in support of ENTR-601-45 at the Muscular Dystrophy Association (MDA) Clinical & Scientific Conference in March 2023.
ENTR-601-45, a proprietary Endosomal Escape Vehicle (EEV™)-conjugated phosphorodiamidate morpholino oligomer (PMO), is the third novel clinical candidate from Entrada’s growing pipeline of EEV-therapeutics. Within Entrada’s growing neuromuscular franchise, each EEV-PMO therapeutic candidate has an oligonucleotide sequence designed and optimized for the specific subpopulation of interest (Figure 2). ENTR-601-45 is designed to address the underlying cause of Duchenne muscular dystrophy due to mutated or missing exons in the DMD gene. ENTR-601-45 is an exon skipping investigational therapy that has the potential to restore the mRNA reading frame and allow for the translation of dystrophin protein that is slightly shortened but still functional.
Figure 1: ENTR-601-45 showed robust exon skipping and dystrophin production in vitro in patient-derived skeletal and cardiac muscle cells
Figure 2: A single IV dose of ENTR-601-45 showed high levels of exon skipping in hDMD mouse skeletal and heart muscle after one week
About Duchenne Muscular Dystrophy
Duchenne muscular dystrophy is a rare, genetic disease that causes progressive muscle degeneration and weakness throughout the body. Duchenne is caused by mutations in the DMD gene, which leads to inadequate production of dystrophin, a protein essential to maintaining the structural integrity and function of muscle cells. Duchenne causes progressive loss of muscle function throughout the body, which limits mobility and causes heart and respiratory complications in the later stages of the disease. Currently approved therapies for Duchenne seek to improve dystrophin production, but to date, the clinical benefits of these products have not been confirmed.About Entrada Therapeutics
Entrada Therapeutics is a biopharmaceutical company aiming to transform the lives of patients by establishing a new class of medicines, Endosomal Escape Vehicle (EEV™)-therapeutics, to engage intracellular targets that have long been considered inaccessible and undruggable. The Company’s EEV therapeutics are designed to enable the efficient intracellular delivery of a wide range of therapeutics into a variety of organs and tissues, resulting in an improved therapeutic index. Through its proprietary, highly versatile and modular EEV platform, Entrada is building a robust development portfolio of oligonucleotide-, antibody- and enzyme-based programs for the potential treatment of neuromuscular diseases, immunology, oncology and diseases of the central nervous system. The Company’s lead oligonucleotide programs include ENTR-601-44 and ENTR-601-45 for the potential treatment of people living with Duchenne who are exon 44 and 45 skipping amenable, respectively, as well as ENTR-701 targeting myotonic dystrophy type 1 (DM1).For more information about Entrada, please visit our website, www.entradatx.com, and follow us on Twitter and LinkedIn.
Forward-Looking Statements
This press release contains forward-looking statements that involve substantial risks and uncertainties. All statements, other than statements of historical facts, contained in this press release, including statements regarding Entrada’s strategy, future operations, prospects and plans, objectives of management, expectations regarding the findings from preclinical data of the Company’s therapeutic candidates, including ENTR-601-45, expectations regarding the continued development and advancement of ENTR-601-44 and ENTR-601-45 for the treatment of Duchenne including the potential benefits they may have, the timing of Entrada's planned regulatory filings regarding its development programs, the ability to develop additional exon skipping programs and the potential therapeutic benefits of its EEV candidates, constitute forward-looking statements within the meaning of The Private Securities Litigation Reform Act of 1995. The words “anticipate,” “believe,” “continue,” “could,” “estimate,” “expect,” “intend,” “may,” “might,” “objective,” “ongoing,” “plan,” “predict,” “project,” “potential,” “should,” or “would,” or the negative of these terms, or other comparable terminology are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. Entrada may not actually achieve the plans, intentions or expectations disclosed in these forward-looking statements, and you should not place undue reliance on these forward-looking statements. Actual results or events could differ materially from the plans, intentions and expectations disclosed in these forward-looking statements as a result of various important factors, including: uncertainties inherent in the identification and development of product candidates, including the conduct of research activities and the initiation and completion of preclinical studies and clinical trials; uncertainties as to the availability and timing of results from preclinical studies; the timing of and Entrada’s ability to submit and obtain regulatory clearance for IND applications and initiate clinical trials; whether results from preclinical studies will be predictive of the results of later preclinical studies and clinical trials; whether Entrada’s cash resources will be sufficient to fund the Company’s foreseeable and unforeseeable operating expenses and capital expenditure requirements; uncertainties associated with the impact of the ongoing COVID-19 pandemic on Entrada’s business and operations; as well as the risks and uncertainties identified in Entrada’s filings with the Securities and Exchange Commission (SEC), including the Company’s most recent Form 10-K and in subsequent filings Entrada may make with the SEC. In addition, the forward-looking statements included in this press release represent Entrada’s views as of the date of this press release. Entrada anticipates that subsequent events and developments will cause its views to change. However, while Entrada may elect to update these forward-looking statements at some point in the future, it specifically disclaims any obligation to do so. These forward-looking statements should not be relied upon as representing Entrada’s views as of any date subsequent to the date of this press release.Entrada Investor/Media Contact
Karla MacDonald
Chief Corporate Affairs Officer
kmacdonald@entradatx.comPhotos accompanying this announcement are available at: https://www.globenewswire.com/NewsRoom/AttachmentNg/ba3f7a21-21c3-4d58-ba0f-3614634a9def
https://www.globenewswire.com/NewsRoom/AttachmentNg/6181a0b4-5fb4-472a-bf41-1a1b32597524